About Us

Who We Are
PROM Health Development (PHD) is a non-profit organization recognized by the Cambodian Royal Government. Based in Phnom Penh, PHD works largely in under served communities throughout Cambodia with the highest burdens of disease. PHD focuses not only on technical and research objectives but on improving the total health and development of communities. PHD supports the Royal Government of Cambodia’s activities particularly in the areas of vector borne and arboviral diseases. After many years of effort, PHD also continues to support the Government in it’s goal to eliminate malaria by 2025. PHD conducts public health outreach and high quality clinical and operational research to inform evidence-based disease and vector elimination efforts. Our founding principles are respect, dignity and compassion for all persons in Cambodia. We achieve success by partnering with local health organizations, NGOs and government agencies to provide high quality health care and research services.
We also partner with local health organizations or government agencies to provide health care services and research including clinical trials by adherence to the principles of good clinical practices (GCPs) and human subject protection (HSP). Our Federal Wide Assurance (FWA) number is FWA00024116.
Our vision is a Cambodia free from malaria and other vector-borne diseases. To get there, we will need to address not only the symptoms, but the root causes of disease.
How We Work
We seek to achieve sustainable gains toward malaria elimination by developing existing local resources and expertise through effective, transparent community engagement. PHD provides technical assistance and builds capacity within communities by working side by side with our partners. Currently PHD has a Memoranda of Understanding with the National Malaria Center, and with Oddor Meanchey Provincial Health Department to provide health care services to both civilian and military personnel. We aim to further expand cooperative health partnerships in Cambodia, seeking to support local efforts where the need is greatest.
We seek solutions which are simple, affordable and practical. Evidence-based clinical research is an important part of our approach, aimed at defining achievable solutions, and tracking progress toward elimination. Our work includes both operational research and clinical trials which adhere to the principles of Good Clinical Practices (GCP) and abide by international norms for human subjects protection. We hold Federal Wide Assurance #FWA00024116. All studies are conducted by our highly experienced field staff in full compliance with local laws and regulations under the supervision of the National Ethics Committee for Health Research (NECRH) in Cambodia. Further details can be found at www.nechr.org.kh. We have also translated key regulatory and ethical training materials into Khmer to better support human subjects protection at www.citiprogram.org.

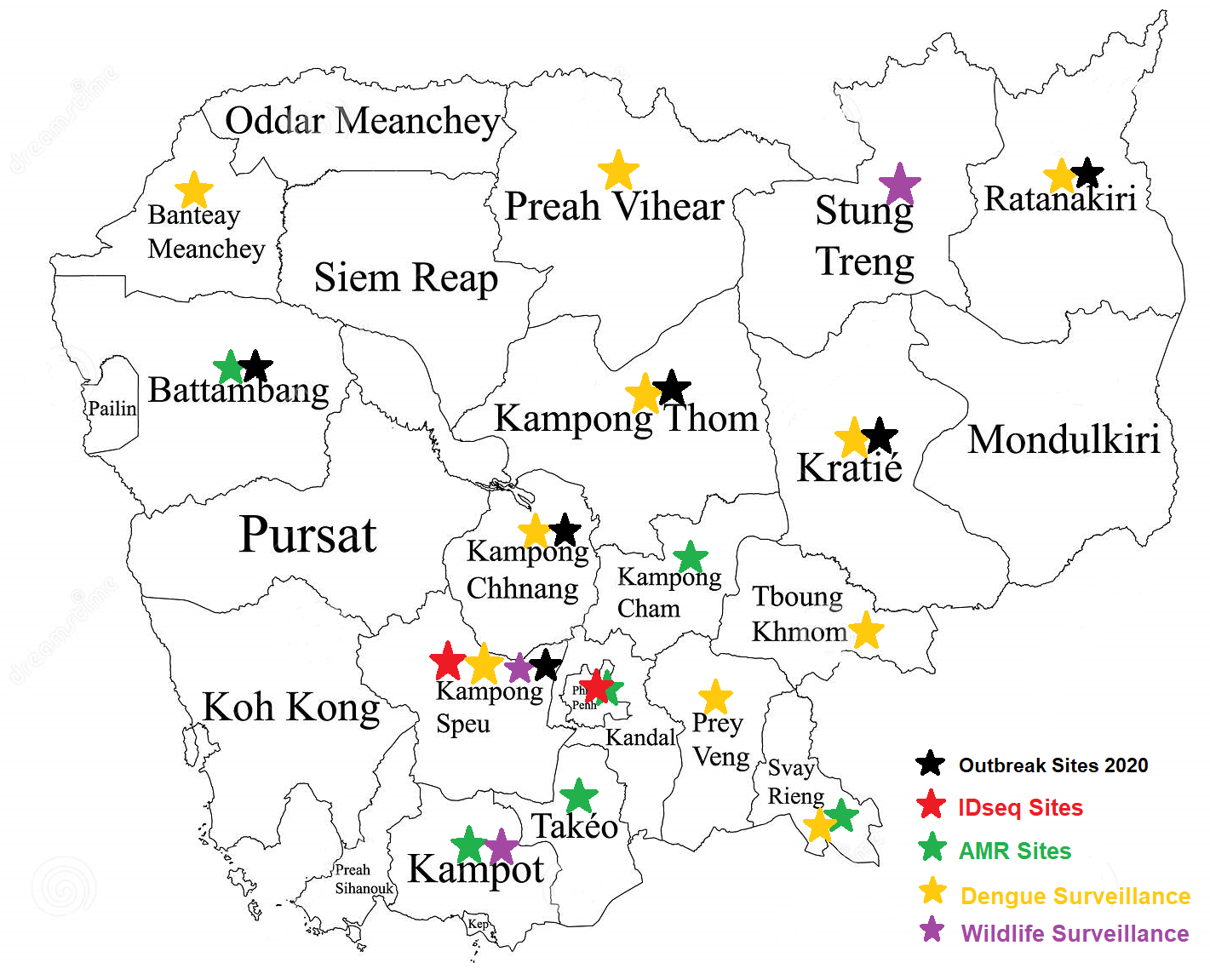
Where We Work
We are committed to working in communities with some of the greatest needs to support research projects. Our team has more than 15 years experience in vector-borne disease control, conducting clinical trials of antimalarials and surveillance of influenza, dengue and diarrheal disease in Cambodia. This includes support for rapid outbreak response to emerging or newly investigated pathogens and antimicrobial resistance in many communities in Cambodia including 4 national hospitals in Phnom Penh city (Detail locations each project in the map).